Generic Sterile Injectable Market Size to Lead USD 124.44 Billion by 2035 Rising Demand for Affordable Injectables and Patent Expirations
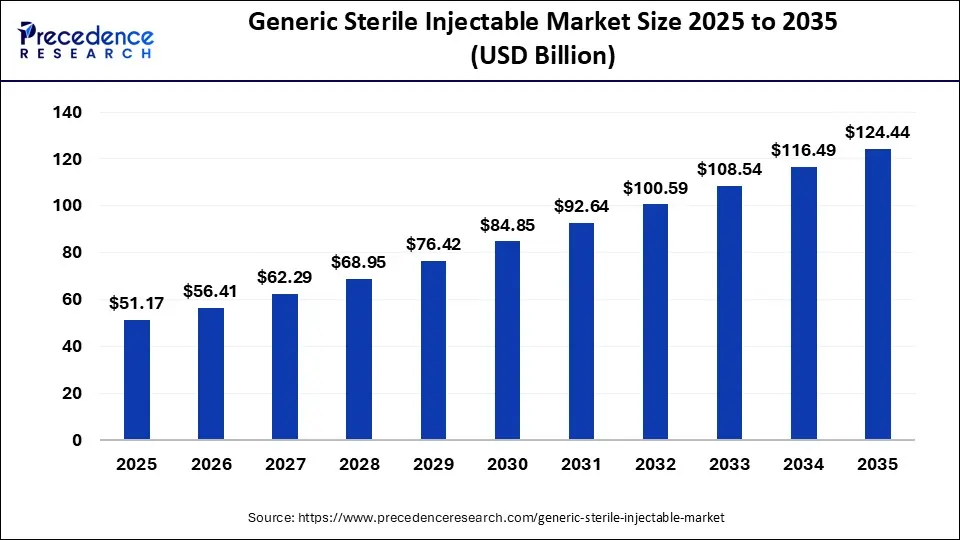
The globe generic sterile injectable market size is expected to be lead USD 124.44 billion by 2035, increasing from USD 51.17 billion in 2025, with a strong CAGR of 9.29% from 2026 to 2035.
Ottawa, April 14, 2026 (GLOBE NEWSWIRE) -- According to Precedence Research, the global generic sterile injectable market size will grow from USD 56.41 billion in 2026 to nearly USD 124.44 billion by 2035, accelerating with a solid CAGR of 9.29% from 2026 to 2035. The generic sterile injectable market is witnessing strong growth, driven by rising chronic disease prevalence, increasing demand for cost-effective therapies, and patent expirations of branded drugs.
The Complete Study is Now Available for Immediate Access | Download the Sample Pages of this Report@ https://www.precedenceresearch.com/sample/1214
Generic Sterile Injectable Market Key Takeaways
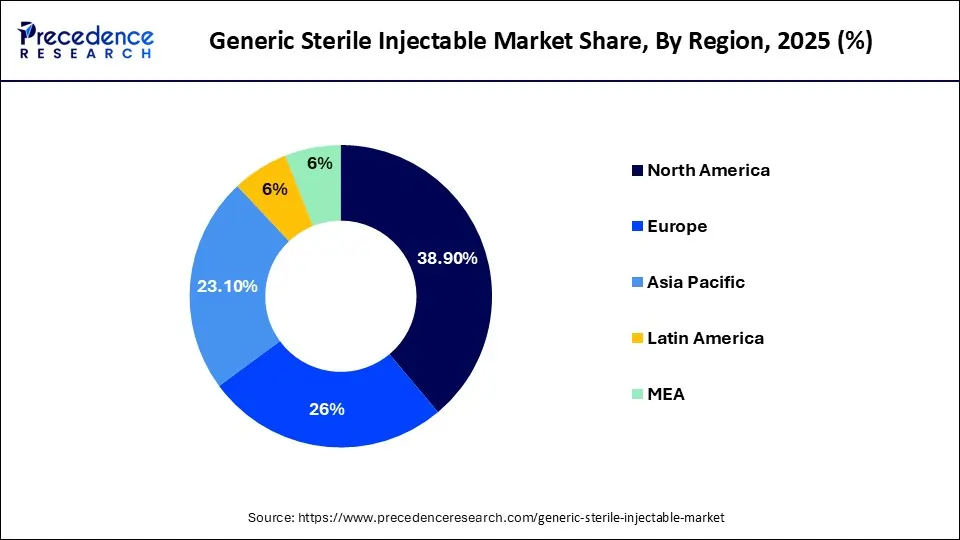
- North America dominated the market with a 38.90% share in 2025.
- The Asia Pacific region is projected to register the fastest growth over the forecast period.
- Based on drug type, the vaccines segment is anticipated to witness the highest growth between 2026 and 2035.
- In terms of therapeutic application, the diabetes segment is expected to grow at the most rapid pace during the forecast period.
- By distribution channel, retail pharmacies are likely to experience the fastest market expansion from 2026 to 2035.
Generic Sterile Injectable Market by Drug Type, 2023-2025 (USD Million)
| Drug Type | 2023 | 2024 | 2025 |
| Monoclonal Antibodies | 14,396.2 | 15,921.4 | 17,643.8 |
| Cytokines | 1,882.0 | 2,077.3 | 2,297.2 |
| Insulin | 6,951.4 | 7,624.4 | 8,377.7 |
| Peptide Hormones | 2,022.3 | 2,202.5 | 2,402.8 |
| Vaccines | 5,173.8 | 5,653.7 | 6,188.9 |
| Immunoglobulin | 1,069.6 | 1,157.4 | 1,254.3 |
| Blood Factors | 1,395.5 | 1,511.9 | 1,640.7 |
| Peptide Antibiotics | 553.1 | 599.9 | 651.8 |
| Others | 8,948.1 | 9,786.5 | 10,722.60 |
Generic Sterile Injectable Market by Therapeutic Application, 2023-2025 (USD Million)
| Therapeutic Application | 2023 | 2024 | 2025 |
| Cancer | 10,872.4 | 12,059.6 | 13,404.0 |
| Diabetes | 7,428.8 | 8,173.0 | 9,008.4 |
| Cardiovascular Disease | 5,885.0 | 6,413.5 | 7,001.5 |
| Central Nervous System | 3,445.0 | 3,740.7 | 4,068.5 |
| Infectious Disorders | 3,739.5 | 4,074.2 | 4,446.3 |
| Musculoskeletal System | 3,283.9 | 3,604.2 | 3,962.9 |
| Others | 7,737.5 | 8,470.0 | 9,288.3 |
Generic Sterile Injectable Market by Distribution Channel, 2023-2025 (USD Million)
| Distribution Channel | 2023 | 2024 | 2025 |
| Hospital Pharmacies | 21,620.7 | 23,703.1 | 26,034.3 |
| Retail Pharmacies | 15,771.4 | 17,370.9 | 19,169.0 |
| Online Pharmacies | 5,000.0 | 5,461.0 | 5,976.7 |
Is Demand for Sterile Injectable Products Driven by Increased Chronic Disease?
Chronic disease is on the rise globally with such diseases as cancer, diabetes, and cardiovascular disease; therefore continued growth of both surgical procedures being conducted and hospital admissions continue to place pressure on product demand. Generic sterile injectables represent a cost-effective alternative to those manufactured by branded manufacturers; thus, they are expected to continue to be adopted widely through healthcare systems throughout the world regardless of developing or developed healthcare practice.
Furthermore, government programs designed to increase the overall level of access to affordable healthcare and increase the available infrastructure within hospitals for providing care are accelerating the overall growth of the sterile injectable market, particularly in developing countries, where providing low-cost healthcare treatment is a critical priority.
➡️ Become a valued research partner with us ☎ https://www.precedenceresearch.com/schedule-meeting
Market Opportunity
The future growth of the generic sterile injectable market will depend on the increasing need for affordable hospital treatment, particularly in oncology, critical care, and biologics support therapies. The growing rate of chronic diseases and the increase in surgical procedures are driving continual demand for ready-to-use injectables. Opportunities for growth in this segment are developing through technological advancements like prefilled syringes, dual-chamber systems, and the use of automation in aseptic manufacturing to improve both safety and efficiency.
Additionally, the expiration of patents for branded injectable drugs offers generic manufacturers the ability to expand their product portfolios. The further growth in this market is fueled by an investment into emerging economies as well as government regulations designed to incentivize domestic manufacturing to ensure supply chain resilience and increased global access to these products.
Built for leaders who move markets. Access live, actionable intelligence with Precedence Q. https://www.precedenceresearch.com/precedenceq/
Regional Insights
Why Is the Chemical Intermediate Market Dominated By North America?
North America currently dominates the global generic sterile injectable market based on its well-established healthcare system, large number of pharmaceutical manufacturers, and high need for drugs provided through hospitals. There are also a significant amount of chronic illnesses in North America, and an increasing number of surgical procedures that will require injectables to treat post-operatively. Generics are encouraged by supportive regulations which facilitate faster regulatory approvals, resulting in greater expansion opportunities. In addition, continued investments in next generation manufacturing technologies, as well as supply chain improvements will help to ensure an uninterrupted supply of sterile injectables throughout North America. The increasing use of biosimilars and cost-effective injectables will also continue to strengthen North America’s market position.
U.S.
The U.S. generic sterile injectable market operates as a hospital-centric, cost-driven pharmaceutical segment characterized by high demand for affordable alternatives to branded injectable drugs. It is largely fueled by the need to reduce healthcare costs, with hospitals and healthcare systems actively procuring generics for critical treatments such as oncology, infections, anesthesia, and emergency care. These products are primarily distributed through hospital pharmacies, which dominate the supply chain due to the complexity and clinical importance of injectable therapies. The market is highly regulated and technologically intensive, requiring strict sterile manufacturing conditions, compliance with U.S. FDA standards, and significant capital investment, which creates strong entry barriers and limits the number of manufacturers.
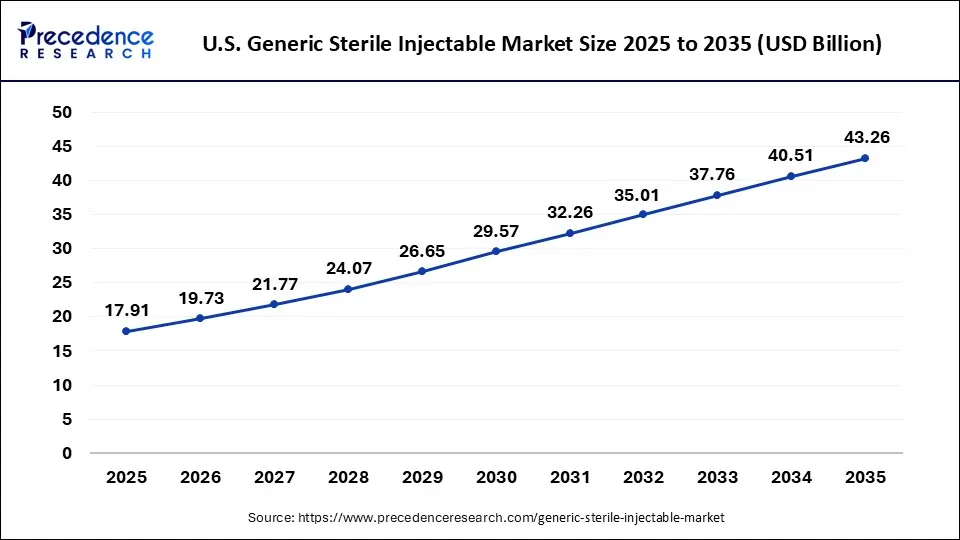
How Big is the Size of U.S. Generic Sterile Injectable Market in 2026?
According to Precedence Research, the U.S. generic sterile injectable market size is valued at USD 19.73 billion in 2026 and is predicted to reach around USD 43.26 billion by 2035, growing at a significant CAGR of 9.22% from 2026 to 2035.
Note: This report is readily available for immediate delivery. We can review it with you in a meeting to ensure data reliability and quality for decision-making.
Try Before You Buy – Get the Sample Report@ https://www.precedenceresearch.com/sample/1214
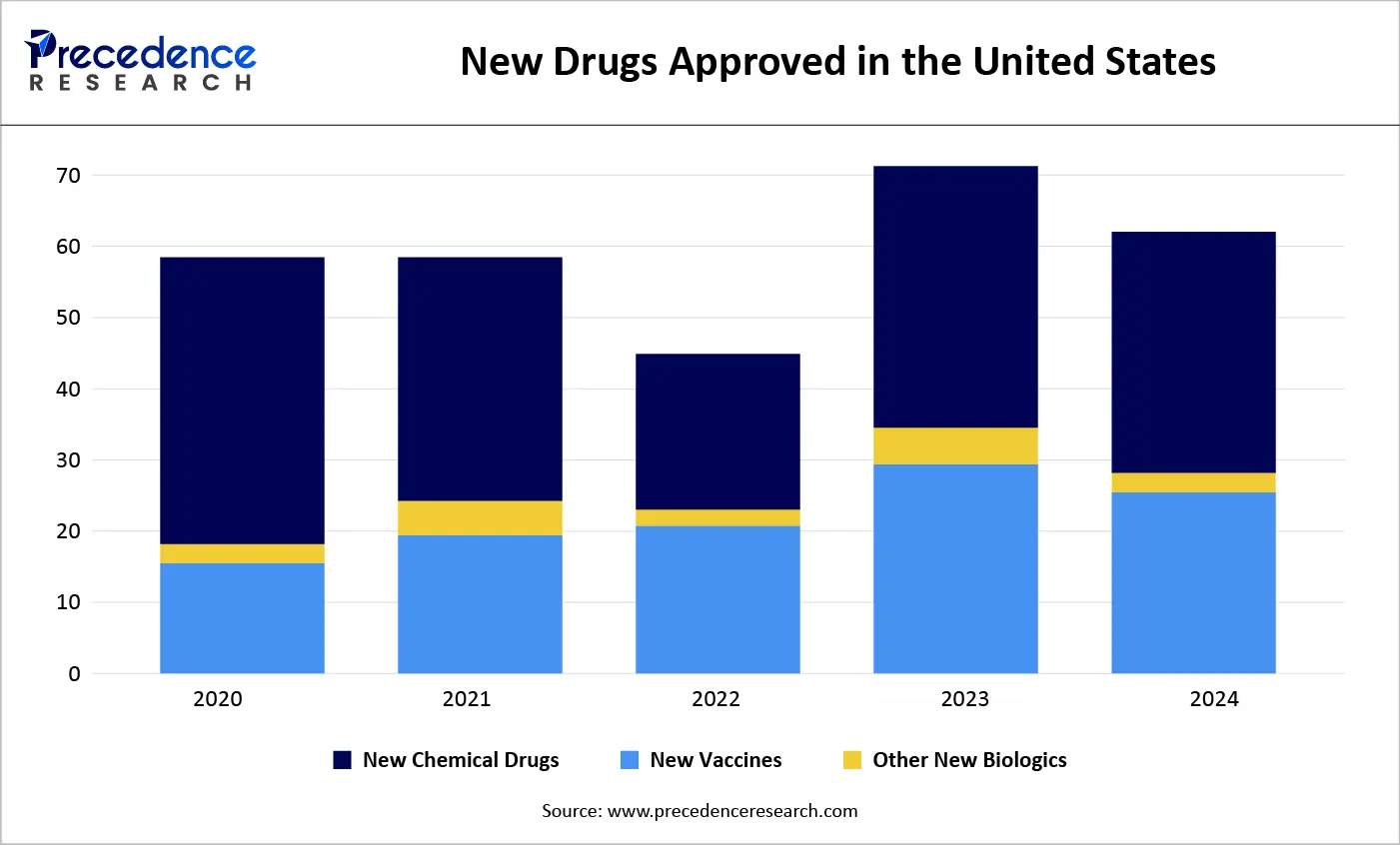
A generic medicine is a drug developed to be equivalent to an already approved medication. Its approval is based on established safety and efficacy data derived from studies of the original drug. Although generic drugs are unbranded, they are identical to their branded counterparts in terms of dosage, administration, and therapeutic effect. According to the U.S. Food and Drug Administration (FDA), a generic drug must contain the same active ingredient and match the brand-name drug in strength, safety, dosage form, route of administration, and performance. Each year, the FDA approves a range of drugs, including vaccines, chemical drugs, and advanced biologics such as gene therapies and antibodies.
How will Asia Pacific Expand Fastest in the Generic Sterile Injectable Market in the Coming Years?
Asia Pacific is the fastest developing region in the global generic sterile injectable market due to growing access to healthcare, an expanding population, and increasing incidence of many chronic diseases. Governments across the region are making large investments toward healthcare systems and incentives to promote the use of lower-cost generic medications. Additionally, Asia Pacific’s low-cost manufacturing capabilities and the presence of global pharmaceutical manufacturing hubs will contribute to growth of the region's generic sterile injectable market. Growing medical tourism and an increasing number of hospital admissions will add additional demand for sterile injectables within this region. Furthermore, favorable regulatory reforms and growing recognition of low-cost treatment options will continue to support growth in the adoption of generic sterile injectable medications throughout the Asia Pacific region.
Get informed with deep-dive intelligence on AI’s market impact https://www.precedenceresearch.com/ai-precedence
Generic Sterile Injectable Market Insights
| Report Metrics | Details |
| Market Size in 2025 | USD 51.17 Billion |
| Market Size in 2026 | USD 56.41 Billion |
| Market Size by 2035 | USD 51.17 Billion |
| Market Growth (2026 – 2035) | 9.29% CAGR |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product, Therapeutic Application, Distribution Channel, Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
➤ Access the Full Generic Sterile Injectable Market Study @ https://www.precedenceresearch.com/generic-sterile-injectable-market
Segmental Insights
Drug Type Insights
What makes Monoclonal Antibodies the Most Dominant Drug Type in 2025?
The monoclonal antibody held dominant market share in 2025. The most commonly demanded type of therapeutic is monoclonal antibody-based treatments. They account for the majority share as they used to treat many complex and chronic illnesses, including cancers and autoimmune disorders. These compounds provide highly targeted therapeutic delivery and unparalleled clinical efficacy, which has improved patient access through an increasing number of biosimilar approvals and hospital reliance on specific, targeted biologics to provide sterile injectables.
Vaccines are among the fastest-growing areas of medicine. Growth is being driven both by increasing global vaccination programmes for people of all ages and an increase in awareness about preventive health. Increased global governments spending on universal vaccination programmes and the emergence of pandemic preparedness plans have provided considerable demand in this area. In addition, new technologies have improved vaccine development methods and new opportunities in adult vaccination markets are likely to further accelerate growth of the vaccine sector.
Therapeutic Application Insights
How did the Cancer Segment Dominate the Generic Sterile Injectable Market in 2025?
Cancer is the most dominant application area because of the high global disease burden associated with cancer, and increasing utilization of injectable cancer therapies, such as chemotherapy, immunotherapy, and targeted biologics. The need for effective and timely delivery of meaningful treatments supports the use of sterile injectables. Ongoing advancement of oncology drugs and increased investment in healthcare will continue to support the leading position of this segment.
Diabetes is expanding rapidly because of the growing global incidence of diabetes, and the increased use of injectable insulin therapies. Patient education, improved diagnosis rates, and increasing access to treatment in developing countries are increasing the demand for insulin therapies for diabetes. Innovations in the formulation and delivery of insulin are further propelling segment growth.
Distribution Channel Insights
Why did the Offline Segment Lead the Generic Sterile Injectable Market in 2025?
Hospital pharmacies dominate the generic sterile injectable market primarily because most sterile injectables are administered in an area requiring professional supervision. The fact that hospital distributions are the primary distribution point confirms that the location requires the immediate administration of the sterile injectable product and that the sterile injectable product must be stored and handled with the utmost care.
Retail pharmacies continue to experience a rapid rate of growth due to the increased demand for convenience of access to medications, particularly with respect to chronic disease management; increased use of outpatient care; and an increasing preference by patients for home-based treatment. In addition, both cold chain logistics and support provided by regulatory agencies will provide continued access to a wider distribution of sterile injectables in retail outlets.
✚ Related Topics You May Find Useful:
➡️ Sterile Injectables CDMO Market: Explore how rising biologics demand and outsourcing trends are accelerating contract manufacturing growth
➡️ Aesthetic Injectables Market: Discover how growing demand for minimally invasive cosmetic procedures is shaping market expansion
➡️ Long-Acting Injectables Market: Understand how sustained-release therapies are improving patient compliance and treatment outcomes
➡️ Sterile Filtration Market: Analyze the increasing need for contamination control in biopharmaceutical manufacturing processes
➡️ Sterile Medical Packaging Market: Gain insights into packaging innovations ensuring product safety and regulatory compliance in healthcare
What are the Major Developments in the Generic Sterile Injectable Market?
- In January 2026, PAI Pharma, U.S.-made generic medicines company acquired Nivagen Pharmaceuticals to expand domestic sterile injectable manufacturing capacity and accelerating development of ready-to-use (RTU) products for the hospital market. (Source- pulse2)
- In July 2026, Cronus Pharma LLC, launched Butorphic® (Butorphanol Tartrate) Sterile Injectable Solution for instant availability in the U.S. animal health market through domestic and regional distributors. (Source – Business Wire India)
Top Companies of the Generic Sterile Injectable Market
- Fresenius Kabi
- Baxter
- Civica
- Pfizer
- Mylan
- Hikma
- Sandoz
- Teva
- Nichi-Iko
- 3M
- Merck & Co., Inc.
- Others
Segments Covered in the Report
By Drug Type
- Monoclonal Antibodies
- Cytokines
- Insulin
- Peptide Hormones
- Vaccines
- Immunoglobulin
- Blood Factors
- Antibiotics
- Others
By Therapeutic Application
- Cancer
- Diabetes
- Cardiovascular Disease
- Central Nervous System
- Infectious Disorders
- Musculoskeletal System
- Others
By Distribution Channel
- Hospitals Pharmacies
- Online Pharmacies
- Retail Pharmacies
- Others
By Geography
- North America
- Asia Pacific
- Europe
- Latin America
- Middle East & Africa
Thank you for reading. You can also get individual chapter-wise sections or region-wise report versions, such as North America, Europe, or Asia Pacific.
Immediate Delivery Available | Buy This Premium Research Report@ https://www.precedenceresearch.com/checkout/1214
You can place an order or ask any questions, please feel free to contact at sales@precedenceresearch.com | +1 804 441 9344
Stay Ahead with Precedence Research Subscriptions
Unlock exclusive access to powerful market intelligence, real-time data, and forward-looking insights, tailored to your business. From trend tracking to competitive analysis, our subscription plans keep you informed, agile, and ahead of the curve.
Browse Our Subscription Plans@ https://www.precedenceresearch.com/get-a-subscription
About Us
Precedence Research is a worldwide market research and consulting organization. We give an unmatched nature of offering to our customers present all around the globe across industry verticals. Precedence Research has expertise in giving deep-dive market insight along with market intelligence to our customers spread crosswise over various undertakings. We are obliged to serve our different client base present over the enterprises of medicinal services, healthcare, innovation, next-gen technologies, semi-conductors, chemicals, automotive, and aerospace & defense, among different ventures present globally.
Web: https://www.precedenceresearch.com
Our Trusted Data Partners:
Towards Healthcare | Towards Packaging | Towards Chem and Materials | Towards FnB | Statifacts | Nova One Advisor | Market Stats Insight
Get Recent News:
https://www.precedenceresearch.com/news
For the Latest Update Follow Us:
LinkedIn | Medium | Facebook | Twitter

Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.

